Clinical Research Team Contact
- Phone: (919)660-2340
- |
- Email: ADRC@duke.edu
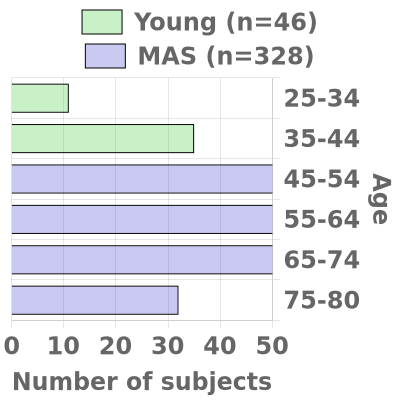
The Duke/UNC ADRC oversees two cohorts, the Memory & Aging Study (MAS) (n=420 over five years) and the Young Cohort (n=120). Participants from both cohorts are recruited from the Triangle and Eastern North Carolina.
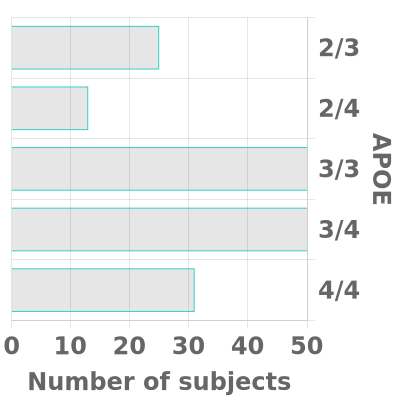
We strive to have substantial rural (~30%) and African-American or other Non-White (~20%) enrollment in both cohorts and our goal is for each cohort to be fairly evenly balanced between men and women. In each cohort, we aim for approximately two-thirds of participants to have a known APOE4 gene and/or a family history of Alzheimer’s disease in a first-degree relative (or in a grandparent, in the case of the Young Cohort).
MAS will develop and maintain a prospectively followed cohort of 420 research participants, ages 45 to 80 and this will enable us to observe key transition periods related to AD risk, including menopause, onset of comorbidities, and the transition to MCI and dementia. To accomplish these goals, the cohort design will collect from two different groups based on affection. The first group will consist of 320 participants who are cognitively normal at entry and roughly equal in age distribution. The second group will include 100 participants symptomatic at entry (MCI or early dementia).
Young Cohort (Ages 25 to 44)
The Young Cohort (n=120) will consist of participants ages 25-44 who will undergo a one-time evaluation and biomarker collection. The availability of this unique comparison group will enable investigators to evaluate the degree to which novel biomarkers or signatures associated with AD in older cohorts are linked to other contributing factors, such as age or genetic risk.
Data and Sample Collection
Each participant contributes UDS-based medical, neurologic, psychiatric, and neuropsychological evaluations.
We also collect blood, urine, and cerebrospinal fluid specimens.
During the baseline visit, we collect motor-sensory assessments, and imaging:
Corrected distance visual acuity on Snellen chart
Zeiss Cirrus HD-5000 AngioPlex retinal and optic nerve imaging device
Participants in the MAS return annually for repeat UDS-based variables, motor-sensory assessments, and blood draw. In the future, they may also be asked to contribute additional MRIs, PET imaging, or biosamples (e.g., urine, stool, cerebrospinal fluid). Each participant is given an adjudicated diagnosis by a consensus panel of clinicians, and this diagnosis is recorded in their study record after each visit. Our goal is to offer all participants the opportunity to enroll for brain donation to the Bryan Brain Bank at the time of their death.